Why Embryo Research?
Assisted reproductive technologies (ARTs) have helped to conceive over 8 million children worldwide and are routinely used in agriculture for animal breeding1,2. However, the success rate of ARTs is low, only 35% even in the best clinics3. One reason for ART failure is that half of human embryos arrest in development during the first week after fertilization4. This phase is usually referred to as "early embryogenesis" or "pre-implantation development" because it occurs before the embryo implants in the uterus, when pregnancy begins. Understanding the basic mechanisms of pre-implantation development is crucial to improving ARTs. On this page, we share our thoughts on why the study of early mammalian embryogenesis is important for our society, as well as being a fascinating research topic for basic researchers.
Important:
We DO NOT carry out research on human embryos, as we use mammalian models, mostly bovine embryos.
We DO NOT sacrifice animals for research, as we generate embryos using material discarded from slaughterhouses and frozen semen.
We DO NOT grow bovine embryos after implantation and we stop their development before organ formation.
Help to conceive
For patients
Infertility is a taboo in our society and is rarely discussed openly. However, over 10% of women and men of reproductive age experience infertility5. ARTs can help these people conceive, but ARTs can also fail, causing emotional and physical stress6 as well as a financial burden7,8. Our research targets basic mechanisms of early embryogenesis with the long-term goal of providing strategies to improve the effectiveness of ARTs.
Aren’t there enough people on the planet already, why do we need to help to conceive?
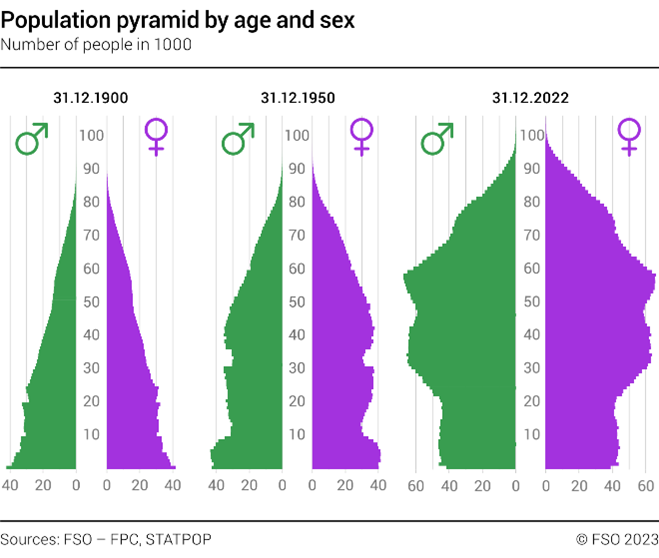
The human population is growing as life expectancy increases worldwide. However, our fertility rate (number of children born per woman) has fallen significantly and our population is ageing9. This is evident in the graphs, which describe the demographic changes in the Swiss population. Reproductive assistance for people who cannot conceive naturally is therefore important not only for their personal lives, but also for population stability.
Preserve endangered species
ARTs are becoming increasingly important for the preservation of endangered species. ARTs allow the cryopreservation of sperm, oocytes, and embryos10. We did not take part in the White Rhino BioRescue project, but we are enthusiastic about it and its potentiality. We hope that our comparative embryogenesis approach will, in the long-term, lead to improved ARTs for endangered species and have a positive impact on the conservation of biological diversity.
Genome editing in embryo research
Genome editing means to changing the DNA content of an organism. In the case of embryos, it refers to modifying the genome of the germ cells or the embryo in its early stages of development, when the embryo has few cells. Genome editing of non-primate embryos has been a routine part of research for many years, allowing the study of many mechanisms of life and disease. Since the emergence of CRISPR technologies, genome editing has become easier, allowing it to be applied to human embryos11,12. Research on human embryos and on editing their genome is a complex and delicate ethical issue. Some countries (e.g. UK and USA) allow it, while others prohibit it (e.g. Switzerland). The International Society for Cell Stem Research has published important guidelines13. In our lab, we DO NOT carry out research on human embryos, but we do use genome editing techniques in other mammalian species, such as bovine embryos.
Are genome editing tools necessary for mammalian embryo research?
We will employ genome editing on early bovine embryos with two main goals.
Firstly, we want to study early development, and genome editing is a powerful tool to modify a gene that we want to study, or to add a fluorescent tag to a protein to visualise embryogenesis under the microscope. These are traditional research approaches in many other model systems, from fruit flies to mouse embryos, which have allowed biological research to make great advances in the last century. Importantly, the genetically modified bovine embryos that we will generate will not be transferred into foster mothers and we will stop their development within 7 days of insemination.
Secondly, we want to know whether genome editing in the germ line and/or embryos, could become a safe technique in ARTs. In principle, preventing the transmission of a genetic disease to one’s offspring would be a key resource for ARTs, giving people with hereditary diseases the chance to have children. We will use our model system to assess whether our genome editing protocols have side effects, such as targeting genomic regions that should not be affected. We will carry out this research in the context of the University Research Priority Program “Human Reproduction Reloaded” (URPP-H2R).
The URPP-H2R seeks to understand and provide policy guidance on the societal impact and legal challenges of rapidly changing medical technologies for human reproduction. The URPP-H2R gives us the opportunity to work in an interdisciplinary context, collaborating not only with leading scientists in the field of genome editing (Gerald Schwank group), but also with philosophers, sociologists, theologists, and lawyers. The meetings of the URPP-H2R are crucial for us, as they allow us to place our research in a wider context and help us to discuss whether modifying the human germline and embryos will ever be ethically justifiable.
References
1. Viana, J. H. 2020 Statistics of embryo production and transfer in domestic farm animals. Embryo Technol. Newsl. 39, (2021).
2. Christian De Geyter. More than 8 million babies born from IVF since the world’s first in 1978. https://www.eshre.eu/Annual-Meeting/Barcelona-2018/ESHRE-2018-Press-releases/De-Geyter (2018).
3. Centers for Disease Control and Prevention. 2017 Assisted Reproductive Technologies - National Summary Report. US Dept Heal. Hum. Serv. 22 (2021).
4. ESHRE Special Interest Group of Embryology & Alpha Scientists in Reproductive Medicine. The Vienna consensus: report of an expert meeting on the development of art laboratory performance indicators†‡. Hum. Reprod. Open 2017, 1–17 (2017).
5. Cox, C. M. et al. Infertility prevalence and the methods of estimation from 1990 to 2021: a systematic review and meta-analysis. Hum. Reprod. Open 2022, 1–24 (2022).
6. Milazzo, A., Mnatzaganian, G., Elshaug, A. G., Hemphill, S. A. & Hiller, J. E. Depression and Anxiety Outcomes Associated with Failed Assisted Reproductive Technologies: A Systematic Review and Meta-Analysis. PLoS One 11, e0165805 (2016).
7. Collins, J. A. An international survey of the health economics of IVF and ICSI. Hum. Reprod. Update 8, 265–277 (2002).
8. Njagi, P. et al. Financial costs of assisted reproductive technology for patients in low- and middle-income countries: a systematic review. Hum. Reprod. Open 2023, (2023).
9. McDonald, P. Low Fertility and the State: The Efficacy of Policy. Popul. Dev. Rev. 32, 485–510 (2006).
10. Hildebrandt, T. B. et al. The ART of bringing extinction to a freeze – History and future of species conservation, exemplified by rhinos. Theriogenology 169, 76–88 (2021).
11. Fogarty, N. M. E. et al. Genome editing reveals a role for OCT4 in human embryogenesis. Nature 550, 67–73 (2017).
12. Zuccaro, M. V. et al. Allele-Specific Chromosome Removal after Cas9 Cleavage in Human Embryos. Cell 1–15 (2020) doi:10.1016/j.cell.2020.10.025.
13. Lovell-Badge, R. et al. ISSCR Guidelines for Stem Cell Research and Clinical Translation: The 2021 update. Stem Cell Reports 16, 1398–1408 (2021).